 
InspireMD's coronary EPS is CE Mark approved. The company notes that in coronary procedures, InspireMD's EPS technology has shown improvements through the MASTER trial findings that revealed a statistically and clinically significant acute advantage of MGuard EPS with regard to ST segment resolution. article: Initial experience with the inspire MD C-Guard stent in the treatment of carotid artery disease - The Journal of Cardiovascular Surgery 2016 June. MGuard is not approved for sale in the United States. 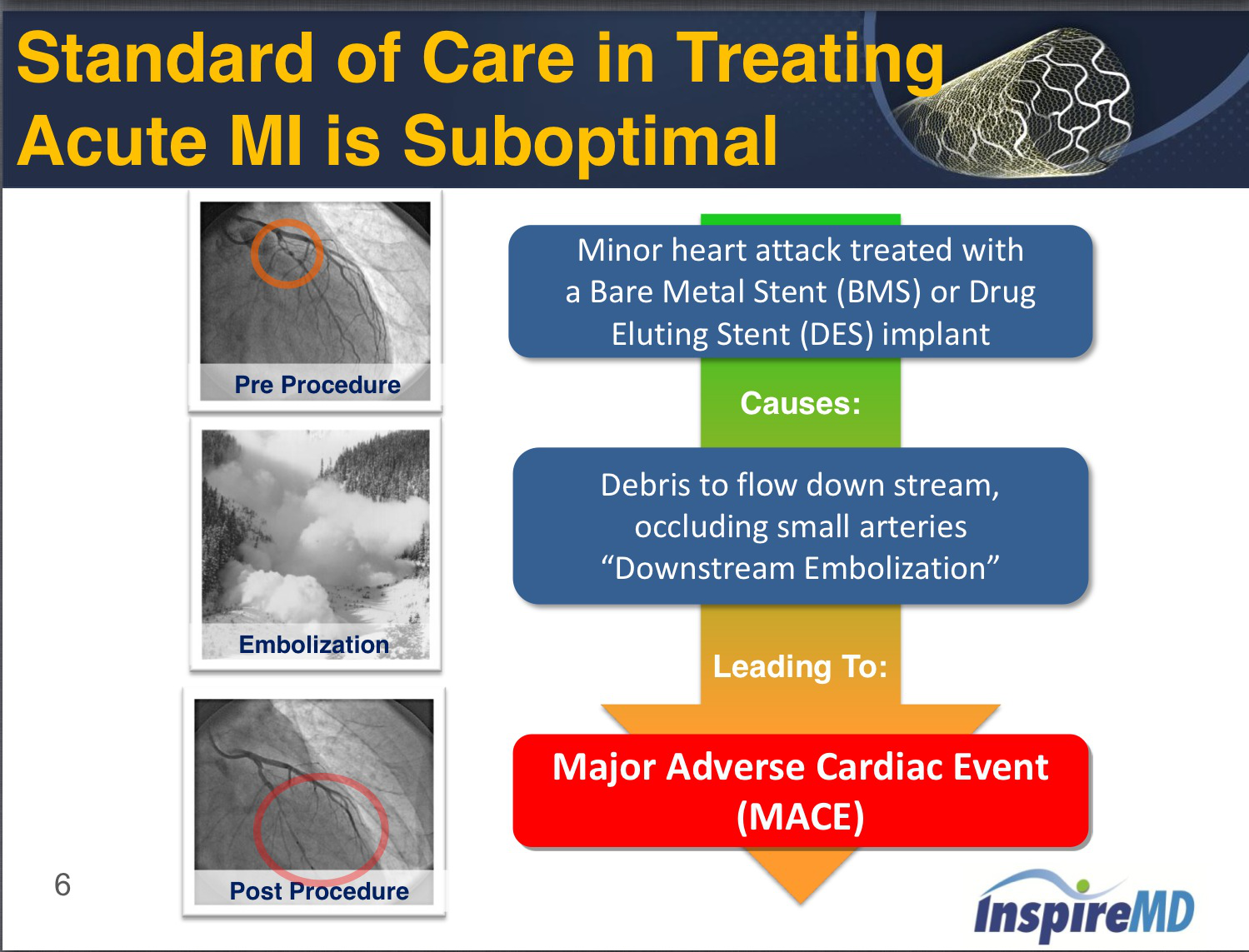
In markets that recognize the CE Mark, the new InspireMD carotid EPS stent will be available in a matrix of sizes ranging from small diameters of 5 X 20 mm to large diameters up to 10 X 60 mm for large carotid arteries. The MicroNet is designed to hold plaque and thrombus in place against the wall of the blocked artery, preventing debris from falling into the bloodstream and causing a potentially fatal downstream blockage or stroke. InspireMD in June began re-introducing the MGuard Prime EPS in Europe after pulling it last April and suspending the Master II trial due over the risk of the stent being dislodged during. The carotid EPS device is based on the company's MicroNet mesh protection platform technology that is also used for the treatment of myocardial infarction patients in InspireMD's commercially available coronary MGuard and MGuard Prime EPS devices.Īccording to the company's press release, the InspireMD carotid EPS is wrapped with a MicroNet mesh to prevent embolic events during and after carotid artery stent procedure. Contact a supplier or the parent company directly to get a quote. (Tel Aviv, Israel) announced that the company received CE Mark approval for its self-expanding nitinol carotid embolic protection stent (EPS). Find out all of the information about the InspireMD product: coronary stent MGuard Prime. Prospective, Randomized, Multicenter Evaluation of a Polyethylene Terephthalate Micronet Mesh-Covered Stent (MGuard) in ST-Segment Elevation Myocardial Infarction: The MASTER Trial.March 15, 2013-InspireMD, Inc. Stone GW, Abizaid A, Silber S, Dizon JM, Merkely B, et al. The products of the company are the CGuard carotid Embolic Prevention System (CGuard EPS) and the MGuard Prime. InspireMD, Ltd. It is focused on the development and commercialization of proprietary MicroNet stent platform technology for the treatment of complex vascular and coronary disease. 0.9%, P=0.0003).Īmong patients with acute STEMI undergoing emergent PCI, MGuard resulted in superior rates of epicardial coronary flow and complete STR, with similar rates of 30 day MACE (1)ġ. InspireMD Inc is the United States based medical device company. 2.3%, P=0.10, respectively) TLR was higher in the MGuard arm (8.6% vs. Baseline characteristics were well matched between the groups. At 12 months, cardiac death and all-cause mortality were numerically lower in the MGuard stent compared to control ((1.0% vs. A total of 433 patients with STEMI presenting within 12 hours of symptom onset undergoing PCI were randomized at 50 sites in 9 countries to the MGuard (n217) or commercially available bare metal or drug-eluting stents (n216). 2.3%, P=0.75) at 30 days were not significantly different between patients randomized to the MGuard stent vs. 1.9%, P=0.06) and major adverse cardiac events (1.8% vs. 82.9%, P=0.006) with comparable rates of myocardial blush grade 2 or 3 (83.9% vs. In addition, the MGuard stent resulted in superior rates of TIMI-3 flow (91.7% vs. The primary endpoint of post-procedure complete STR was significantly improved in patients randomized to the MGuard stent compared to control (57.8% vs. 
The infarct vessel was the left anterior descending coronary artery in 40.2% of patients, the right coronary artery in 51.3%, and the left circumflex coronary artery in 8.3%. InspireMD (OTC:NSPR) said it issued a voluntary field action after receiving reports that its MGuard Prime EPS stent can dislodge during implantation procedures. Median age was 59 years and 24% of patients were female. The objective of the MASTER trial was to demonstrate the superiority of the MGuardTM/MGuard Prime stent over commercially-approved bare-metal (BMS) or drug-eluting stents (DES) in achieving better myocardial reperfusion in primary angioplasty for the treatment of acute ST-elevation myocardial infarction (STEMI).Ī total of 433 patients with STEMI presenting within 12 hours of symptom onset undergoing PCI were randomized at 50 sites in 9 countries to the MGuard (n=217) or commercially available bare metal or drug-eluting stents (n=216).īaseline characteristics were well matched between the groups. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
